
How to cite this article:
KACHI Science and Communication. Rare Diseases: What Are Their Peculiarities for Pharmaceutical Marketing? KACHI Scientific Communication and Pharmaceutical Marketing [Internet]. 2025. Available at: https://kachi.com.br/blog-conteudo-cientifico-doenca-rara/
Contextualization of Rare Diseases
In 2025, the World Health Organization (WHO) classified rare diseases as “a global health priority for equity and inclusion” and reinforced the recommendation that countries develop national strategies to address this issue in a more coordinated way (WHO 2025). This initiative is timely: it is estimated that up to 300 million people are affected worldwide, across more than 7,000 different conditions (NIH 2022; WHO 2025).

Although “rare,” they are not so distant from everyday medical practice. For example, the National Organization for Rare Disorders (NORD) reports that 1 in every 10 Americans lives with a rare disease — about 30 million people in the U.S. alone (NORD 2022). In Europe, initiatives such as Orphanet map thousands of low-prevalence pathologies, gathering data on incidence, prevalence, and treatments — although nearly 95% of them still lack approved therapies (NORD 2022).
What is a rare disease?
In Brazil, the National Health Surveillance Agency (Anvisa) defines a rare disease as one that affects up to 65 people per 100,000 inhabitants (Ministério da Saúde 2020; RDC nº 205/2017). Although the definition is numerical, these diseases share common challenges: they are generally severe, chronic, and progressive, significantly impacting patients’ and families’ quality of life (Caffrey 2021 BMJ).

Challenges of Rare Diseases
In England, a recent study analyzed medical records from 58 million people and identified more than 300 rare diseases, corresponding to about 1.5% of the population studied. On average, these patients take more than five years to receive an accurate diagnosis and also face a shortage of specialists sufficiently familiar with each condition (Thygesen 2025).
The picture is complex, but there are signs of progress. Since the 1980s—especially after the approval of the Orphan Drug Act in the United States—research and development of orphan drugs have gained momentum, driven by incentives designed to stimulate the launch of therapies for very small populations (NORD 2022; FDA 2023).

At a global scale, collaboration among laboratories, healthcare professionals, academic researchers, regulators, and patient associations has been encouraged by organizations such as the National Institutes of Health (NIH) and the European Medicines Agency (EMA), with the goal of identifying urgent clinical needs and expediting the availability of therapeutic solutions (NIH 2022; NICE 2022; Rambaran 2024 JAMA).
With this focus, in 2017 Anvisa published a resolution to accelerate research and regulatory approval of new therapies aimed at these conditions—therapies capable of positively impacting the lives of patients and families. Even so, challenges such as early diagnosis and the complexity of approving new therapeutic options remain (RDC nº 205/2017).
In parallel with advances in research, the WHO and NIH emphasize the importance of providing comprehensive and humanized care for these individuals, including:
- medical awareness campaigns,
- early diagnosis programs,
- psychosocial support,
- creation of multidisciplinary reference centers,
- and care protocols (WHO 2025; NIH 2022).
In addition, it is worth highlighting the generation of real-world evidence to demonstrate clinical and economic benefits, which is particularly relevant for price and reimbursement negotiations in public and private healthcare systems (Kesselheim 2019 N Engl J Med).
This set of actions is essential so that those living with these conditions are not, literally, “too rare to matter,” but are recognized and included in public health policies.
Thus, although they are called “rare,” these diseases represent a vast and nuanced universe. As the regulatory and scientific landscape shows, each step in structuring clear national strategies and partnerships among various stakeholders can represent a major leap in the quality of life of those with a chronic, complex condition often invisible to the rest of society.
The Pharmaceutical Market and Marketing Challenges in Rare Diseases
Even with the expansion of incentive policies and growing global attention to rare diseases, the pharmaceutical market still faces considerable barriers to launching and sustaining products in these areas (WHO 2025). On one hand, there is fertile ground for innovation and highly specialized therapies; on the other, regulatory and economic uncertainties prevail, influencing companies’ strategic planning.
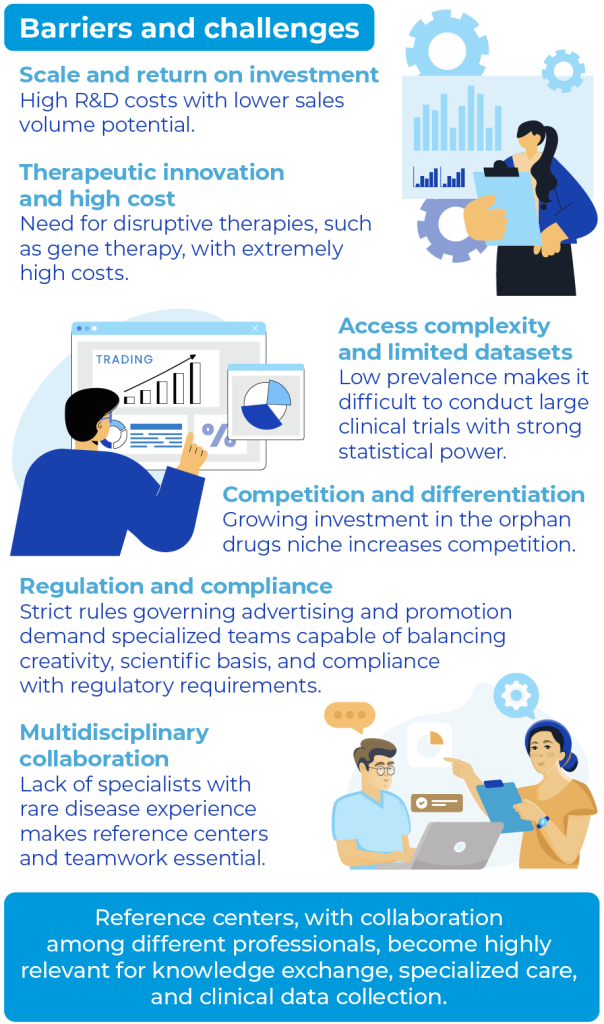

Scale and Return on Investment
Drugs intended for rare diseases, called orphan drugs (NORD 2022), carry risks associated with lower returns on investment: the industry must balance high research and development (R&D) costs with potentially smaller sales. This justifies the adoption of incentives such as extended market exclusivity, tax benefits, and registration facilitation, as in the U.S. Orphan Drug Act and Brazil’s RDC nº 205/2017 (FDA 2023; RDC nº 205/2017).
Therapeutic Innovation and High Cost
A distinctive feature of rare diseases is the possibility of disruptive therapies, such as gene therapies or next-generation biologics. Although they open new market opportunities, these solutions demand substantial R&D investments, resulting in treatment costs that may exceed millions of dollars per year (Kesselheim 2019 N Engl J Med). This scenario puts pressure on health systems, which must assess cost-effectiveness and sustainability, as well as on pharmaceutical companies, which need to justify high prices with data on efficacy and clinical impact (NICE 2022).
Access Complexity and Scarcity of Extensive Data
Another challenge is market access, as price and reimbursement negotiations with payers (public or private) depend on robust data that demonstrate clear clinical value. In many cases, the low incidence of the disease makes it difficult to conduct extensive clinical trials, reducing the statistical power of the research (Thygesen 2025). At the same time, regulatory agencies such as EMA and Anvisa require long-term safety and efficacy evidence, particularly since these are often unprecedented therapies (RDC nº 205/2017).
Competition and Differentiation
Even though the patient population is smaller, more and more companies are investing in this niche, intensifying competition. Differentiation strategies — whether through additional clinical benefits, new formulations, or patient support programs — become crucial to demonstrate that a drug offers more than what already exists (NORD 2022).
Regulation and Compliance
In several countries, including Brazil, there are strict rules governing drug advertising and promotion, both for the general public and healthcare professionals (Mailankody 2017). Examples include the ban on direct-to-consumer (DTC) advertising in some markets and the requirement for educational materials to undergo a rigorous review process. The purpose of these laws and guidelines is to preserve patient safety, the accuracy of information, and company transparency. Consequently, companies must adopt robust compliance practices, creating communication materials that meet all legal requirements — a process that often demands specialized teams capable of balancing creativity, scientific rigor, and regulatory conformity.
However, this set of rules and successive approvals can impose delays or restrictions on marketing and communication strategies, especially in the case of orphan drugs, where there is urgency to make therapies available. Thus, even with incentive policies, the path to marketing treatments for rare diseases demands carefully developed tactics that ensure clarity of information and compliance with ethical and legal standards (Mailankody 2017; RDC nº 205/2017).
Multidisciplinary Collaboration
The shortage of specialists with experience in rare diseases is one of the biggest obstacles to adequate diagnosis, treatment, and follow-up of these patients. Therefore, collaboration among different professionals becomes indispensable (NIH 2022; Thygesen 2025).
The creation of reference centers, composed of multidisciplinary teams — clinicians, geneticists, neurologists, pharmacists, nurses, therapists, and other professionals — promotes knowledge exchange and strengthens the continuum of care. These centers also serve as hubs for training, specialized care, and collection of relevant clinical data (WHO 2025; NIH 2022).
Partnerships among stakeholders
Partnerships among stakeholders—such as reference centers, patient associations, medical organizations, and researchers—are essential in the rare-disease context to:
- Build scientific credibility around the therapeutic value of new treatments, connecting technical knowledge to patients’ real lives, providing clinical evidence to support regulatory and reimbursement decisions, and strengthening trust in new therapies. (NIH 2022; Rambaran 2024 JAMA; Kesselheim 2019 N Engl J Med).
- Support the design and conduct of multicenter clinical trials. (Thygesen 2025),
- Optimize the generation of real-world evidence, helping to demonstrate clinical benefits in settings outside controlled trials. (FDA 2023; NICE 2022)
- Encourage adoption of innovative treatments in clinical practice by engaging specialists and integrating new therapies into care protocols.(WHO 2025)
- Strengthen arguments before HTA bodies and payers, demonstrating the clinical, economic, and social value of therapies; facilitating regulatory approval and national registration as well as reimbursement negotiations and incorporation of treatments for low-prevalence diseases—thereby helping reduce access inequalities. (Kesselheim 2019 N Engl J Med; NORD 2022; WHO 2025; NICE 2022).
Thus, the pharmaceutical market for rare diseases balances high innovation potential with uncertain returns, demanding marketing approaches guided by:
- solid scientific evidence;
- strategic partnerships;
- clear market-access strategies;
- effective communication that amplifies the needs of people living with rare diseases and demonstrates each new therapy’s relevance and differentiation.

Value Approach and Differentiation in Rare Diseases
To stand out in a market as specific as that of rare diseases, companies must clearly and objectively demonstrate the clinical and social value their product offers (Kesselheim 2019 N Engl J Med). This includes arguments that justify not only the efficacy and safety of the medicine, but also its impact on the quality of life of patients and families — many of whom often have no other treatment options available.
Building Value Arguments
In general, the development and commercialization costs for these therapies are high, which is reflected in elevated prices. Therefore, manufacturers must support their value propositions with robust data and methodologies recognized by evaluating agencies (NICE 2022). Examples of tools that legitimize results include:
- Multicenter clinical trials;
- Collection of real-world evidence;
- Measurement of quality-of-life indicators.
Use of Economic and Social Data
To convince payers, health-system managers, and government agencies to approve reimbursement or incorporation of a drug, it is essential to include:
- Cost-effectiveness analyses
- Cost-utility assessments
- Long-term morbidity reduction projections (Thygesen 2025)
In addition, psychosocial impacts — such as reducing caregiver burden and improving patients’ ability to attend school or work — can be decisive differentiators in well-supported dossiers (Rambaran 2024 JAMA).
Creating Narratives that Engage Stakeholders
An important part of differentiation also lies in how pharmaceutical companies communicate their product. It is common to use narratives that highlight the stories of real patients, illustrating how the new therapy transforms lives (NORD 2022). These stories, combined with clinical data, strengthen connections with patient associations, healthcare professionals, and regulators.
Synergy with Patient Associations and Specialized Centers
Another fundamental strategy is close collaboration with patient associations, medical societies, and reference centers, joining forces to develop clinical protocols and usage guidelines. This relationship not only enhances the scientific legitimacy of the therapy but also increases awareness among medical and public communities (NIH 2022). Ultimately, this synergy facilitates drug adoption in the market and consolidates the manufacturer’s reputation as a responsible ally in the fight against rare diseases.
Therefore, the value approach and differentiation in rare diseases cannot be limited to marketing slogans or a technical dossier. It is necessary to combine robust evidence, empathetic narratives, and strategic partnerships, showing that the company goes beyond delivering a product: it is committed to offering real solutions for a group of patients who, historically, have had fewer treatment opportunities.
Market Access and Reimbursement Policies in Rare Diseases
In the rare disease landscape, achieving market access — that is, ensuring that the drug is incorporated into healthcare systems and made available to patients — is one of the greatest challenges (Thygesen 2025). The high cost of orphan therapies, stemming from the complexity of research and development, raises budgetary concerns for both public and private payers (Kesselheim 2019 N Engl J Med).
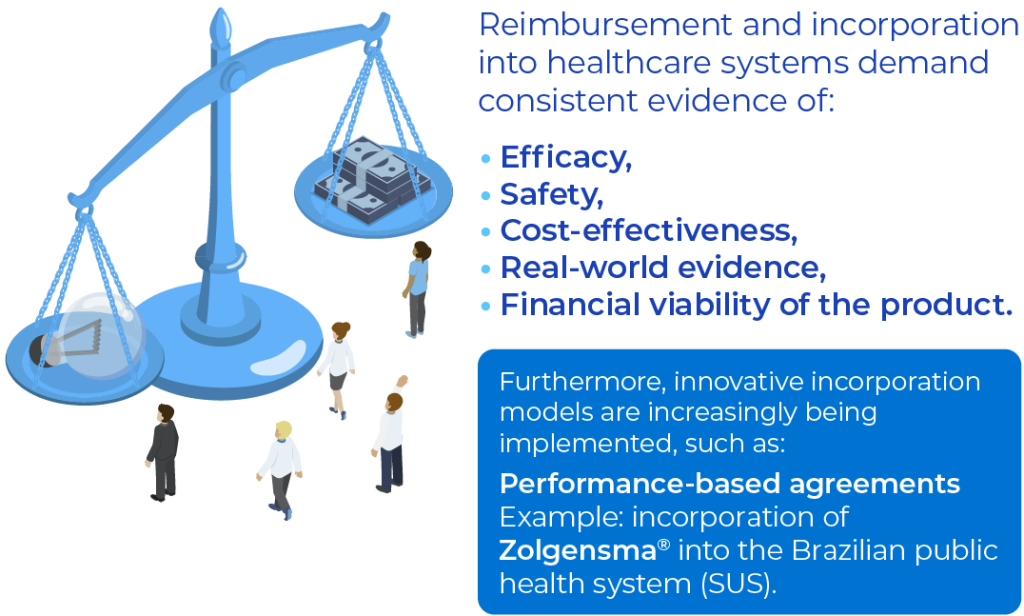
Access Dossiers and Price Negotiations
In many markets, there are multiple payers (private insurers, public systems, among others), each with their own evaluation criteria and processes (NORD 2022). In addition, many governments enforce spending caps and budget containment policies, which require robust evidence that high prices are justified by clinical and social benefits (NICE 2022).
Thus, in incorporation or coverage negotiations, pharmaceutical companies must present solid data on efficacy, safety, cost-effectiveness, and financial viability, demonstrating the value of the therapy in contexts where few or no treatment options exist (Kesselheim 2019 N Engl J Med; NICE 2022; NIH 2022). Even when a medicine is recognized as “orphan,” additional requirements may arise to ensure that its high cost is proportional to the benefits offered. In this process, the use of real-world evidence has gained relevance, as it reinforces clinical outcomes and effectiveness in broader populations and everyday practice settings (FDA 2023).
Expanded Access and Compassionate Use Regulations
To reinforce value arguments and broaden market penetration, many pharmaceutical companies provide the therapy free of charge, even before formal approval, through expanded access programs and compassionate use mechanisms (RDC nº 38/2013). This ensures that patients without therapeutic alternatives are not left unattended during regulatory review.
In addition, after registration, some companies maintain patient support programs to help ensure correct use of the drug and facilitate collection of additional clinical information (Rambaran 2024 JAMA). These mechanisms also allow healthcare professionals to become familiar with the treatment and its monitoring in daily practice.
Example of Risk-Sharing: Zolgensma in SUS
In certain cases, technology incorporation involves performance-based agreements, where payment is conditioned on the clinical outcomes achieved. An example of this approach was adopted in March 2025, when Brazil incorporated Zolgensma® for the treatment of Spinal Muscular Atrophy (SMA) type 1 into the public health system (Ministério da Saúde 2024). The Ministry of Health began making payments as patients reached specific motor milestones. This reduces financial risk for the health system while requiring the manufacturer to provide practical proof of the drug’s effectiveness.
Outcome: Aligning Value and Viability
For orphan drugs to effectively achieve market access, it is crucial to adopt a strategic approach that combines consistent clinical and pharmacoeconomic evidence, strong partnerships with health systems, and flexible negotiation models — such as outcome-based payment, as exemplified by the Zolgensma® case (Ministério da Saúde 2024).
When well executed, these strategies allow innovative therapies to reach those who need them most, transforming the prognosis of diseases that previously lacked treatment options (Thygesen 2025).
Communication Strategies with Stakeholders: Engagement and Continuing Medical Education
In the world of rare diseases, effective communication with stakeholders is crucial to:
- raise awareness,
- accelerate diagnoses, and
- strengthen access to innovative therapies.
This section addresses two essential fronts: the engagement of patient associations and advocacy groups, and continuing medical education (CME) targeted both at reference centers and Key Opinion Leaders (KOLs), as well as at primary care professionals.

Engagement of Patient Associations and Advocacy Groups
Collaboration with patient associations is highly productive in the development of disease awareness campaigns and also in engaging stakeholders in decisions about purchasing or prescribing orphan therapies (Rambaran 2024 JAMA).
Patient associations and advocacy groups play a fundamental role in giving voice to those living with rare diseases. These organizations provide direct support to patients and families, share real experiences, and influence the healthcare policy agenda. By collaborating with them, pharmaceutical companies can:
- improve their communication strategies,
- validate the value of new therapies, and
- strengthen their arguments in negotiations with payers (Rambaran 2024 JAMA; NORD 2022).
Continuing Medical Education (CME)
Due to the low prevalence and complexity of rare diseases, many healthcare professionals struggle to stay updated on the signs, diagnostics, and treatments of these conditions. Investing in CME is therefore essential to improve care.
To deepen investigations, confirm diagnoses, and manage therapies, the focus must be on reference centers and KOLs, who concentrate expertise and disseminate the latest scientific evidence (NIH 2022; NICE 2022; Rambaran 2024 JAMA).
However, it is equally important to train primary care professionals, who are usually the first to identify signs suggestive of a rare disease. Studies indicate that many patients wait more than five years for an accurate diagnosis, largely due to the lack of specific knowledge among these physicians (Thygesen 2025; NIH 2022). Targeted initiatives for front-line professionals can accelerate referrals to specialized centers, contributing to earlier diagnoses and better prognoses.
Integration of Engagement and Education
The combination of patient-association engagement with a robust CME strategy forms an important pillar for transforming the rare-disease landscape. This integrated approach enriches medical practice, accelerates knowledge dissemination, and strengthens market-access negotiations, facilitating access to therapies that can radically change patients’ lives.
How a Specialized Agency Can Leverage Results
A specialized agency, such as KACHI, can support the transformation of pharmaceutical market challenges in rare diseases into opportunities for growth and consolidation through an integrated approach that encompasses scientific communication, multichannel strategies, relationship management, and market access consulting.
Scientific Communication Consulting
Producing clear, rigorous, and audience-adapted educational materials is essential to translate complex information into accessible messages. By developing content that respects compliance standards and current legislation, the agency ensures that messages convey credibility and safety both to healthcare professionals and to the general public. This type of consulting not only strengthens the pharmaceutical company’s image but also contributes to the dissemination of scientific knowledge — which is fundamental for advances in the diagnosis and treatment of rare diseases.
Multichannel Strategies
To expand the reach of messages, KACHI develops marketing campaigns and plans with tailored, integrated actions across in-person and digital channels. Examples include:
- social media campaigns,
- email marketing,
- support for the creation of print collateral, and
- organization of in-person events.
This multichannel approach ensures consistent, audience-specific communication, delivering information effectively and on time, maximizing engagement, and strengthening brand presence in the market.
Relationship Management
Building solid partnerships is essential for success in the rare-disease segment. KACHI coordinates collaboration with patient associations, Key Opinion Leaders (KOLs), and medical societies, providing support in advisory boards and collaborative projects. For example:
- producing reprints signed by KOLs,
- implementing initiatives with Digital Opinion Leaders (DOLs).
These strategies strengthen relationship networks, expand knowledge dissemination, and increase the credibility of new therapies among stakeholders.
Market Access Consulting
KACHI also plays a role in building value and differentiation arguments, which are essential for negotiations with payers. By analyzing data on efficacy, safety, cost-effectiveness, and budgetary viability, the agency helps prepare robust dossiers that demonstrate the real benefits of therapies.
This strategic consulting ensures that arguments are aligned with healthcare system requirements, contributing to reimbursement approvals and the incorporation of drugs into the market.
By combining scientific communication consulting, multichannel strategies, relationship management, and market access consulting, a specialized agency like KACHI drives results, strengthens the image of pharmaceutical companies, and above all, ensures that innovative therapies reach those who truly need them.

Conclusion
Opportunities and Responsibility
In a market as challenging as that of rare diseases, the success of a product goes beyond sales, reflecting its real and transformative impact on patients’ lives. Adopting an ethical, patient-centered approach is essential to ensure that innovative therapies deliver tangible benefits, improving the quality of life of those who face long journeys to obtain a diagnosis and access to effective treatments.
This responsibility extends from proving efficacy and safety to implementing market access strategies based on robust evidence and real-world data (Kesselheim 2019 N Engl J Med; NICE 2022; NIH 2022).
In this scenario, a specialized agency such as KACHI plays a decisive role in orchestrating the execution of integrated strategies in communication, continuing medical education, and stakeholder relationship management. By combining scientific consulting, multichannel campaigns, and strategic partnerships with patient associations, reference centers, and Key Opinion Leaders (KOLs), the agency adds value to pharmaceutical companies and the healthcare system, ensuring that high-complexity therapies reach those who need them most in a sustainable and ethical way (Rambaran 2024 JAMA; RDC nº 38/2013).
Thus, transforming challenges into opportunities requires the creation of an environment where each health investment not only generates financial return but, above all, promotes a significant improvement in patients’ lives, reaffirming the commitment to social responsibility and innovation.
Bibliographic references
- Caffrey L. Rare diseases: bridging the knowledge gap. BMJ. 2021;374:n2032.
- FDA. Orphan Drug Act & Related Regulations. FDA.gov; 2023.
- Kesselheim AS. Drug Development for Rare Diseases in the Era of Precision Medicine. N Engl J Med. 2019;381(3):103–105.
- Mailankody S. Pharmaceutical Marketing for Rare Diseases. JAMA. 2017;317(10).
- Ministério da Saúde. Doenças Raras — Informações Gerais [Internet]. Agência Nacional de Vigilância Sanitária (ANVISA); 2020.
- Ministério da Saúde. Incorporação de Zolgensma ao SUS [Internet]. 2024.
- NICE. Guidance on Rare Diseases [Internet]. NICE.org.uk; 2022.
- NIH. Rare Disease Day [Internet]. NIH; 2022.
- NORD. NORD 2022 Impact Report [Internet]. 2022.
- Rambaran S. Trust in Clinical Practice: A Systematic Review. JAMA. 2024;12:21–29.
- RDC nº 38/2013. Agência Nacional de Vigilância Sanitária; 2013.
- RDC nº 205/2017. Agência Nacional de Vigilância Sanitária; 2017.
- Schwartz LM. Medical Marketing in the United States, 1997–2016. JAMA. 2019;321(1):80–96.
- Thygesen JH, et al. Prevalence and demographics of 331 rare diseases and associated COVID-19-related mortality among 58 million individuals: a nationwide retrospective observational study. Lancet Digit Health. 2025;7:e145–56.
- WHO. Rare diseases: a global health priority for equity and inclusion [Internet]. 2025.



