
Introduction
For more than two decades, the pharmaceutical industry has invested heavily in the collection and management of scientific and commercial data. From clinical trial registries to real-world evidence platforms, the sector has built vast repositories of information. However, the growing complexity of healthcare markets, regulatory environments, and therapeutic innovation is pushing companies to evolve: the emphasis is no longer only on data generation but on decision-making. In this emerging paradigm, scientific communication plays a pivotal role, transforming static information into actionable insights for healthcare professionals (HCPs) and patients.1

From Data Abundance to Decision Urgency
The volume of biomedical data has expanded exponentially, with PubMed alone indexing more than one million new articles annually.2 While this explosion of knowledge represents progress, it also creates cognitive and operational overload. Healthcare professionals need timely, relevant, and contextualized information to guide treatment choices. Pharmaceutical companies, in turn, must transform data into narratives that enable decisions.
Real-time data use, supported by artificial intelligence (AI), is shortening the feedback loop from weeks to hours, enabling commercial and scientific teams to adjust strategies instantly. This marks a shift from a passive era of “data storage” to an active era of “decision execution”.1
Foundations of the Decision-Making Era
Big Data is one of the factors that has accelerated this transition. Initially described by the “3 Vs” — Volume, Variety, and Velocity — the concept has now expanded to encompass seven Vs:3
- Volume
- Variety
- Velocity
- Veracity
- Visualization
- Variability
- Value
These dimensions underline not only the scale and diversity of data but also the challenges of ensuring its reliability, interpretability, and actionable relevance.4,5
Business analytics further demonstrates how raw data can be transformed into decisions across three analytical levels:6
- Descriptive analytics (summarizing historical data to understand performance)
- Predictive analytics (using statistical models and machine learning to forecast future outcomes)
- Prescriptive analytics (evaluating scenarios and recommending optimal actions)
Together, these stages build the foundation for evidence-based decision-making.

The Role of Scientific Communication
Emerging trends in scientific communication illustrate how data and decision-making intersect. The rise of digital dissemination — blogs, wikis, social media, and preprints — has accelerated the flow of knowledge, though concerns about peer review and data quality remain.7,8
At the same time, AI-driven personalization allows highly tailored content delivery in pharmaceutical and healthcare marketing, shifting the focus from brand-centric to customer-centric communication.9–11 The industry is also moving from multichannel to omnichannel engagement, integrating real-time feedback to optimize the HCP journey.9
Alongside these advances, transparency and trust remain central. Ethical issues such as data privacy, algorithmic bias, and informed consent are shaping the boundaries of responsible innovation.5,10,12
So, effective scientific communication is becoming the bridge between evidence and action. For pharma marketers, this means:
- Building content hubs that function as decision-support tools.1,6
- Delivering omnichannel campaigns that align timing, channel, and message.9,13
- Embedding transparency in AI-assisted materials to build HCP trust.5,14
Key trends accelerating this shift include real-time engagement through AI-powered analytics, contextualization of evidence into decision-support formats (infographics, clinical pathways, and comparative data), personalized omnichannel strategies tailored to HCP needs, and the growing integration of scientific and commercial narratives.1,13–15
Why Promotion Remains Relevant: Awareness, Memory, and Action
A key condition for decision-making is awareness. For someone to be interested in a topic, they must first know about it, at least have heard of it, and be able to recall it.2 In pharmaceutical contexts, this principle translates into brand awareness, memory, and recall shaping physician prescribing behavior.
- Awareness: Physicians are unlikely to prescribe or recommend a product they have not encountered. Awareness is the gateway to trial and adoption.2
- Memory and Recall: Repetition and reinforcement strengthen recall, which is a strong predictor of marketing effectiveness. Strategies such as cross-channel exposure and storytelling enhance memorability.13
- Availability Bias: Cognitive science shows that physicians tend to choose products that come most easily to mind. Pharmaceutical promotion ensures that brands remain in the “evoked set” of options.2
- Trust and Quality: Familiarity lowers perceived risk and increases confidence. Brands that are known and remembered are more likely to be trusted.2
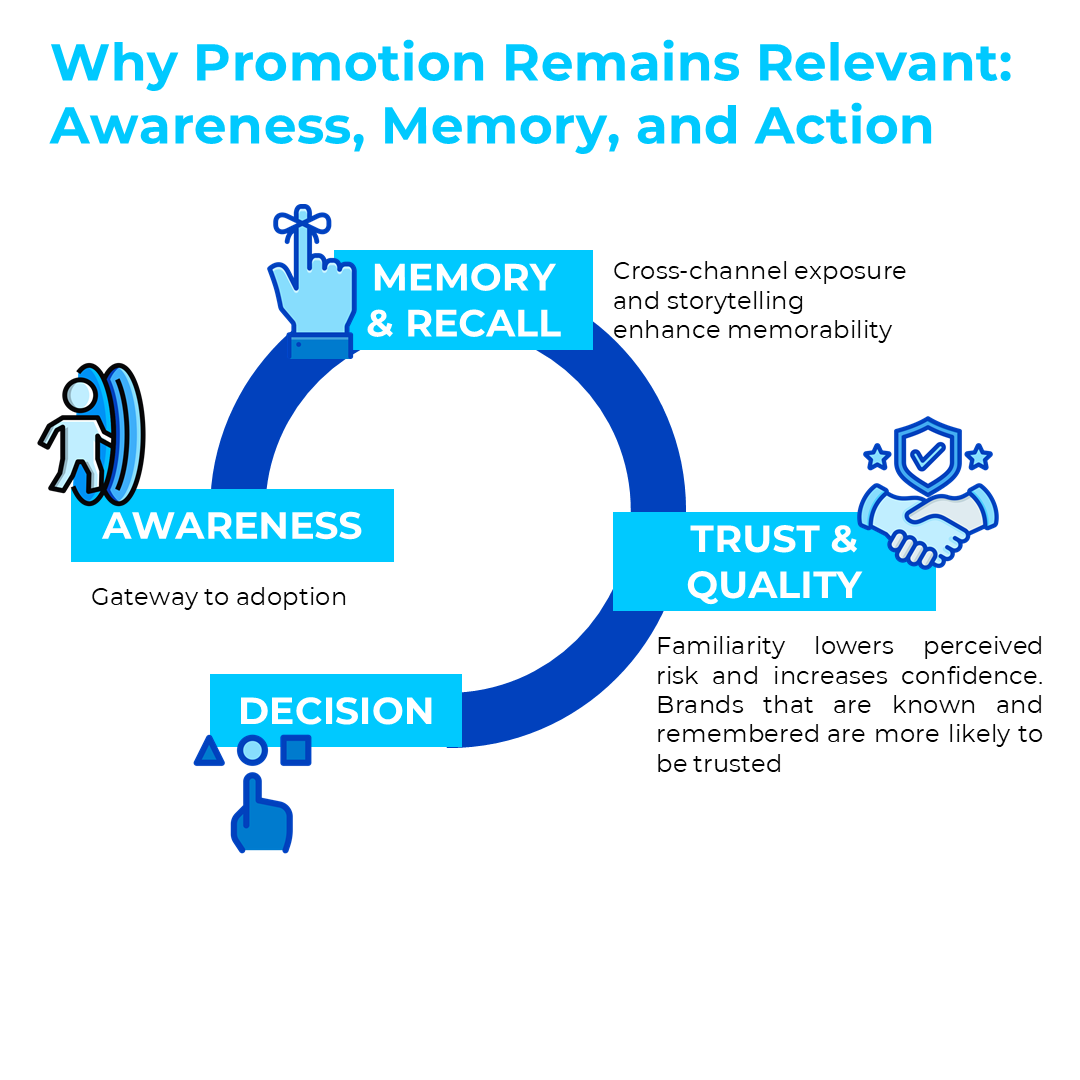
Thus, even in a decision-driven era, pharma promotion remains essential: it sustains awareness, reinforces memory, and ensures that scientific information and brand value are part of the decision-making process.
Implications for Pharma Marketing
In this decision-making era, speed, relevance, and trust become the critical differentiators. Companies must ensure:
- Data quality and governance: Decisions are only as strong as the data that support them. Rigorous validation and transparency build HCP trust .16
- Human-centered communication: While AI automates analysis, the physician–patient–industry dialogue must remain empathetic and credible.17
- Actionable outcomes: The ultimate measure of communication is whether it enables better decisions—improved prescribing, faster adoption of innovations, and clearer patient conversations.1

Conclusion
The pharmaceutical industry is experiencing a profound transformation: the transition from the data era to the decision-making era. In this new landscape, scientific communication is not about volume but about value. Companies that succeed will be those capable of converting evidence into decisions, accelerating innovation, and reinforcing trust with healthcare professionals.1
The message is clear: data alone is no longer enough; decisions are the new currency of scientific communication, and promotion remains the gateway to those decisions.2
—
How to cite this article:
KACHI. Título. São Paulo: KACHI Comunicação Científica, Dia Mês Ano. Disponível em: https://kachi.com.br/en/trends-in-health-communication-insights-for-modern-pharma-marketing
References:
- IQVIA. The Crucial Role of Real-Time Data Use in Pharma Marketing [Internet] [Internet]. IQVIA Blogs. 2024;Available from: https://www.iqvia.com/blogs/2024/11/the-crucial-role-of-real-time-data-use-in-pharma-marketin
- Schwartz LM, Woloshin S. Medical Marketing in the United States, 1997-2016. JAMA 2019;321(1):80–96.
- Ooi PTB, Yuan NK. Ethical use of Big Data and Artificial Intelligence in Biomedical Research [Internet]. The Bioethics Advisory Committee Singapure. 2025, Feb;Available from: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://file.go.gov.sg/bac-bdai-report-2025.pdf
- Floridi L. Big Data and Their Epistemological Challenge. Philosophy & Technology 2012;25(4):435–7.
- Ooi PTB, Yuan NK. Ethical use of Big Data and Artificial Intelligence in Biomedical Research [Internet]. The Bioethics Advisory Committee Singapure. 2025, Feb;Available from: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://file.go.gov.sg/bac-bdai-report-2025.pdf
- Ibeh CV, Asuzu OF, Olorunsogo T, Elufioye OA, Nduubuisi NL, Daraojimba AI. Business analytics and decision science: A review of techniques in strategic business decision making. World Journal of Advanced Research and Reviews 2024;21(2):1761–9.
- Christie AP, White TB, Martin PA, et al. Reducing publication delay to improve the efficiency and impact of conservation science. PeerJ 2021;9:e12245.
- Korjonen MH. Clinical trial information: developing an effective model of dissemination and a framework to improve transparency [dissertation] [Internet]. University College London. 2011, Jun;Available from: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://discovery.ucl.ac.uk/id/eprint/1344051/1/1344051.pdf
- Pi T, Ruan H, Xie Y, Hypothesis N. Content Management Strategies for Pharmaceutical Companies Navigating Omnichannel Marketing [Internet]. 2024 [cited 2025 Sep 4];Available from: https://www.lek.com/insights/hea/cn/sr/deep-dive-content-management-strategies-pharma
- Dingre S, Girbane V, Sankhe N, Todakari P, Borawake D, Deshmukh M. Emerging trends in pharmaceutical marketing strategies for allopathic drugs. International Journal of Science and Research Archive 2025;15(3):667–73.
- Bates A. New business models are the medicine needed to save pharma from the disease of disruption [Internet]. Eularis – Accelerating Business Growth. 2023 [cited 2025 Sep 4];Available from: https://eularis.com/new-business-models-are-the-medicine-needed-to-save-pharma-from-the-disease-of-disruption/
- Cardona-Acevedo S, Agudelo-Ceballos E, Arango-Botero D, et al. Applications of Machine Learning (ML) in the context of marketing: a bibliometric approach. F1000Res 2025;14:92.
- Mor J, Kaur T, Menkes DB, Peter E, Grundy Q. Pharmaceutical industry promotional activities on social media: a scoping review. J Pharm Health Serv Res 2024;15(4):rmae022.
- Bromme R, Mede NG, Thomm E, Kremer B, Ziegler R. An anchor in troubled times: Trust in science before and within the COVID-19 pandemic. PLoS One 2022;17(2):e0262823.
- Orzan G, Zara IA, Purcarea VL. Neuromarketing techniques in pharmaceutical drugs advertising. A discussion and agenda for future research. J Med Life 2012;5(4):428–32.
- Yu JH, Lee S, Kim YJ, Kim WY, Lee MJ, Kim Y. Assessing Post-Marketing Requirements for Orphan Drugs: A Cross-Sectional Analysis of FDA and EMA Oversight. Clin Pharmacol Ther 2024;116(6):1560–71.
- Thorp HH. Trust edges up-slightly. Science 2024;386(6724):831.



